If you have high Lp(a), what should you do? The answer to that question is about to change. Four new medications that lower Lp(a) by up to 94% are in clinical trials: pelecarsen, olpasiran, lepodisiran, and muvalaplin. The first cardiovascular outcomes data is expected in 2026.
Lp(a) is the strongest genetic risk factor for heart disease. About 20% of people have elevated levels, and current medications (statins, ezetimibe) do not lower it. These four trials will answer the question every cardiologist is asking: does lowering Lp(a) actually prevent heart attacks and strokes?
In this post, we’ll walk through each of the four drugs (pelacarsen, olpasiran, lepodisiran, and muvalaplin), the trial behind each one, its timeline, and what the data shows so far.
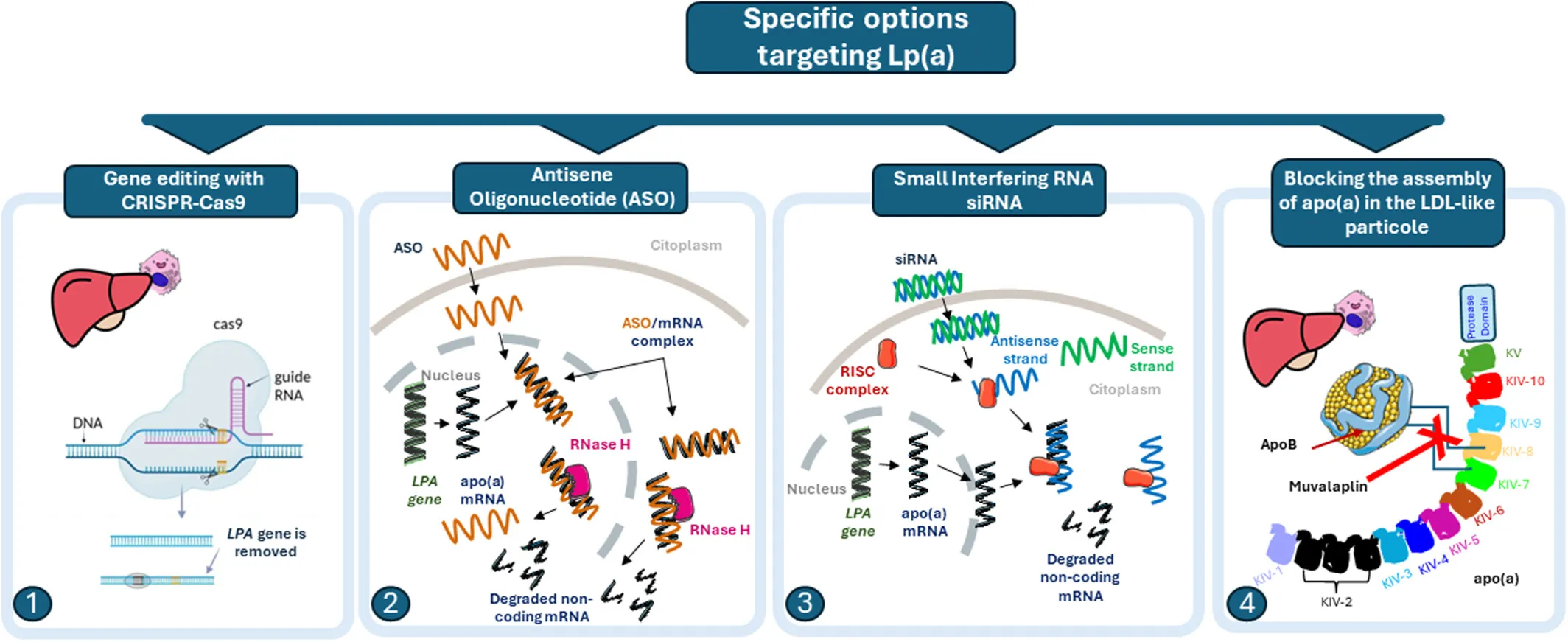 Diagram of various potential approaches to lowering Lp(a).
Diagram of various potential approaches to lowering Lp(a).
Pelacarsen (Novartis)
How it works: Pelacarsen is an antisense oligonucleotide (ASO) that blocks production of apolipoprotein(a) in the liver. It is given as a monthly subcutaneous injection (80 mg).
The Lp(a) HORIZON trial sarted in December 2019 and enrolled 8,323 patients with established cardiovascular disease and Lp(a) of at least 70 mg/dL. Enrollment completed in 2023. Novartis announced Phase 3 data is expected in the first half of 2026, with regulatory submissions planned for the second half. Phase 2 trials showed pelacarsen lowered Lp(a) by up to 80%. Lp(a) HORIZON is the first cardiovascular outcomes trial for any Lp(a)-lowering therapy, so its readout will define the field.
Olpasiran (Amgen)
Trial:
Olpasiran is a small interfering RNA (siRNA) therapy that silences the LPA gene in the liver. It is given as a injection every 12 weeks.
The Phase 3 trial started in December 2022 and enrolled roughly 7,000 patients with established ASCVD and Lp(a) of at least 200 nmol/L. Estimated completion is December 2026. In the earlier Phase 2 OCEAN(a)-DOSE trial, olpasiran lowered Lp(a) by more than 95% at the highest dose.
Lepodisiran (Eli Lilly)
Trial:
Lepodisiran is an siRNA therapy targeting apolipoprotein(a). Phase 2 data suggests dosing may be as infrequent as every 6 months.
TThe ACCLAIM-Lp(a) trial started in 2024 and is still enrolling. It plans to randomize 16,700 patients, making it the largest of the four trials. It enrolls both secondary prevention (established ASCVD) and primary prevention (high-risk first-event) patients. Estimated completion is 2029. The Phase 2 ALPACA trial showed a 93.9% reduction in Lp(a) at the highest dose.
Muvalaplin (Eli Lilly)
Muvalaplin is the only oral drug in the group. It is a small molecule that blocks the assembly of the Lp(a) particle by disrupting the bond between apolipoprotein(a) and apolipoprotein B.
*KRAKEN (Phase 2) ran through 2024 and showed reductions of 47-86%, depending on the dose. Lilly’s Phase 3 cardiovascular outcomes trial is now enrolling, with results expected later this decade.
Lp(a) lowering drugs compared
| Drug | Sponsor | Modality | Dosing | Outcomes trial | Started | Expected results |
|---|---|---|---|---|---|---|
| Pelacarsen | Novartis / Ionis | Antisense oligonucleotide | Monthly injection | Lp(a) HORIZON (n=8,323) | Dec 2019 | H1 2026 |
| Olpasiran | Amgen | siRNA | Injection every 12 weeks | OCEAN(a)-Outcomes (n=~7,000) | Dec 2022 | Dec 2026 |
| Lepodisiran | Eli Lilly | siRNA | Injection every 6 months | ACCLAIM-Lp(a) (n=16,700) | 2024 | 2029 |
| Muvalaplin | Eli Lilly | Oral small molecule | Daily pill | Phase 3 (enrolling) | 2025 | Late decade |
When will Lp(a) drugs be available?
The Lp(a) HORIZON readout later this year will be the first time we know whether lowering Lp(a) translates into fewer heart attacks and strokes. If it succeeds, it will validate the entire class and likely accelerate approvals for the other three drugs.
In the meantime, the 2026 AHA/ACC guidelines recommend that every adult test their Lp(a) at least once. Knowing your Lp(a) now means you will be ready to act the moment these therapies reach the market.
Get your free 30-day heart health guide
Evidence-based steps to optimize your heart health.
